'C' denotes the concentration of the dissolved gas. Where, 'P' denotes the partial pressure of the gas in the atmosphere above the liquid. The mathematical formula of Henry's law is given by: P ∝ C (or) P = k H.C. Importance of Henry's Constant :To increase the solubility of CO2 in soft drink and soda water, the bottle is sealed under high pressure. In chemistry, Henry's law is a gas law that states that the amount of dissolved gas is proportional to its partial pressure in the gas phase. It was formulated by William Henry in 1803.īesides, what is the significance of Henry's law constant KH? Values for Henry's law constants are expressed in units of atmospheres for air to moles per cubic meter for water (atm-m 3/mol) or in a dimensionless unit described as K H ′ = K H/(RT) where K H ′ is the dimensionless Henry's law constant, K H is the Henry's law constant (atm-m 3/mol), R is the ideal gas constant (8.20575 × 10Īdditionally, what does Henry's law state? Henry's law states that at a constant temperature, the amount of a gas that dissolves in a liquid is directly proportional to the partial pressure of that gas in equilibrium with that liquid.
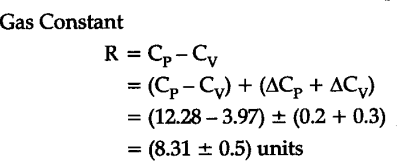
Similarly, what are the units for Henry's law constant? N = 2 mol, T = 300 K, P = 2.50 x 10 5 Nm - 2 R = 8.(k) is Henry's law constant (often in units of M/atm) Calculate the volume occupied by 2 moles of an ideal gas atĢ.5 x 10 5 Nm - 2 pressure and 300 K temperature. We are given P 1 = 740 mm Hg P 2 = 760 mm Hg How much volume will it occupy at S.T.P.? Solution About 200 cm 3 of a gas is confined in a vessel at 20☌ and 740 mm Hg pressure. SolutionĪccording to ideal gas equation, PV = nRT Calculate the number of moles of hydrogen (H 2 ) present in a 500 cm 3 sample of hydrogen gas at a pressure of 760 mm of Hg and 27☌. = 8.314 J mol - 1 K - 1 The value of Gas constant R in different unitsĥ. In the SI system of units, under NTP conditions, P = 101325 N m - 2 Since, 4.182 x 10 7 erg = 1calorie Hence, Normal pressure, P = (1 x 76 x 13.6 x 981) dyne cm - 2 Molar volume under normal temperature and pressure If 'R' is to be expressed in CGS units, the unit of pressure should be dyne cm - 2 and volume in cm 3 mol - 1. ) When pressure is expressed in atmosphere, and volume in mL Under standard condition of temperature and pressure i.e., when P = 1 atm Conditions and problems I)When pressure is expressed in atmosphere and volume in litres The magnitude and unit of 'R' depends upon the units in which pressure, volume and temperature are expressed. The universal gas constant is a measure of energy change (work done) per mole of the gas for one degree change in its temperature. The word 'ideal' is used here because in reality no gas obeys the above condition and the gases, which deviate from ideality are called as real gases. PV = RT (Ideal Gas Equation) The Ideal Gas Equation is also known as the equation of state for gases as it expresses the quantitative relation ship between the four variables that describe the state of the gas. Therefore the Ideal gas Equation is derived as: However, the numerical value of 'R' varies with the units in which pressure and volume are expressed. Where 'R' is the universal gas constant of proportionality. This means that 'K' is directly proportional to the number of moles, 'n', i.e., The volume of a gas is directly proportional to the number of moles of gas at constant temperature and pressure (Avogadro's law). The numerical value of the constant of proportionality (K) depends upon the quantity of gas. The above relationship is very useful for converting the volume of a gas from one set of conditions to another. Check Other Dimensional Formulas: Dimensions of Newton Dimensions of Wavelength Dimensions of. Therefore, the Universal Gas Constant is dimensionally represented as M 1 L 2 T-2 K-1. V 2 is the volume of same amount of gas at temperature T 2 and pressure P 2, then Universal Gas Constant Presure × Volume × n × Temperature-1. Now, if V 1 is the volume of a gas at temperature T 1 and pressure P 1. The gas constant is, by convention, symbolized R. A simple and direct method of deriving this equation is as follows:Īccording to Boyle's law, for a given mass of a gas at constant temperature gas constant (universal molar gas constant): The gas constant, also known as the universal molar gas constant, is a physical constant that appears in an equation defining the behavior of a gas under theoretically ideal conditions. This is known as combined Ideal Gas Equation.

By combining Boyle's and Charles' laws, an equation can be derived that gives the simultaneous effect of the changes of pressure and temperature on the volume of the gas.
